The May issue of Nature Medicine reports what I am pretty sure was the first covid challenge trial. It was a small one, designed to track how the viral load develops after infection, to see how quickly tests detect infection, and to check procedures to pave the way for subsequent challenge trials. Recall that a challenge trial, also called a human infection trial, is one in which the participants are deliberately exposed to the disease, and then reside in the hospital under close medical observation and care.
Killingley, B., Mann, A.J., Kalinova, M., Boyers, A., Goonawardane, N., Zhou, J., Lindsell, K., Hare, S.S., Brown, J., Frise, R. and Smith, E., 2022. Safety, tolerability and viral kinetics during SARS-CoV-2 human challenge in young adults. Nature Medicine, 28(5), May, 1031-1041.
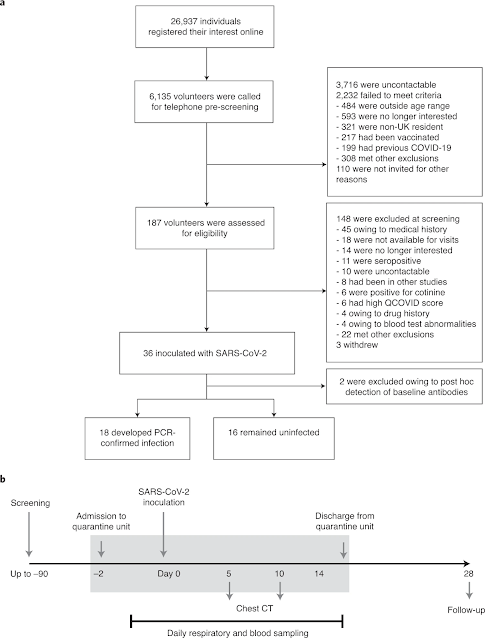
"36 volunteers aged 18–29 years without evidence of previous infection or vaccination were inoculated with 10 TCID50 of a wild-type virus (SARS-CoV-2/human/GBR/484861/2020) intranasally in an open-label, non-randomized study (ClinicalTrials.gov identifier NCT04865237; funder, UK Vaccine Taskforce). After inoculation, participants were housed in a high-containment quarantine unit, with 24-hour close medical monitoring and full access to higher-level clinical care."
...
"Written informed consent was obtained from all volunteers before screening and study enrollment. Participants were given a donation of up to £4,565 to compensate for the time and inconvenience of taking part in the study (including at least a 17-day quarantine). This was calculated using the National Institute for Health Research (NIHR) formula and the UK national living wage."
Two of the volunteers were found to have previous antibodies, and of the remaining 34, 53% (18 people) were infected with the disease after 5 days.
Here's a schematic of how the trial proceeded, starting with almost 27,000 people who volunteered online to participate in the trial, from which the final 36 participants were chosen.
In contrast, the Phase 3 clinical trial of the Pfizer vaccine had almost 22,000 people in the vaccine group and in the placebo group, and reported after four months that only 8 participants in the vaccinated group had contracted the disease, compared to 162 in the placebo group. So the vaccine was 95% effective (only 5% of the 170 infections were in the vaccinated group).
Big (Phase 3) vaccine trials aren't comparable to small preliminary trials, so my point here is just that in the challenge trial the rate of infection of unvaccinated volunteers in 5 days is more than fifty times that rate over four months in the traditional clinical trial, in which participants go about their lives and get infected by chance. That's one of the reasons that challenge trials offer the possibility of fast and efficient testing.
***********
When the study was completed, in 2021, the NY Times published a column with the headline
Britain Infected Volunteers With the Coronavirus. Why Won’t the U.S.?, By Kate Murphy Oct. 14, 2021
It ended with the following paragraph:
"As one participant in Britain’s Covid human challenge trial put it: “You know the phrase ‘one interesting fact about yourself’ that strikes terror into everyone? That’s now solved forever. I did something that made a difference.”
***********
Of course challenge trials are controversial in lots of ways. See some discussion here of the ethics of paying challenge participants.
No comments:
Post a Comment
Note: Only a member of this blog may post a comment.